You are herehalting rusty metal_2
halting rusty metal_2
I have a theory about painting and preserving rusted steel; that goes against some common ideas. I've been told that once steel is rusted, no amount of sanding or coating will stop the rust, it will come back. Rusted steel is ferrous oxide, or hydrated ferrous oxide, which I think means that the source of oxygen is water. My thinking is that if you can stop oxygen from contacting the steel, then the rust will stop.
Now for an observation, to lead toward my theory of why some coatings fail over rusted steel. Years ago, I worked in an old wooden building, which was painted white with a blue trim. Not just any blue trim, but mega-chroma blue mixed with white. If it were a tertiary blue, like Cape Cod blue, that would have been nice, in just about any tint. But this particular blue had to go. The wooden trim was unusual, in that it was mounted at an angle. It was tilted toward the ground, not the sky. A key note here is that it had lots of little dried half bubbles, little craters that were bubbles put in at the time of the previous painting. I applied a coat of paint, and went to do the next area. Upon looking at what I had just painted, I noticed that the round rims of the little craters were now sticking out of my freshly applied coat of paint. My reaction was to apply more paint to the same area. A few minutes later, the craters were back. I applied paint a third time, and again, they returned. This amount of paint was enough to cause some serious runs, so more wasn't an option.
This was baffling, since gravity should have pulled the paint down and away from the board, and made it gather in a way that covered the craters. Static electricity can cause paint to have strange effects when applying it to sheets of plastic. But this was wood, not plastic, old wood that had been there for years. I didn't use enough friction to cause a static charge when cleaning the wood. All I could figure was that it was atmospheric pressure which forced the paint back against the board. So I think that when a paint is applied to a rusty surface, most of that paint gathers on the low spots of the rough surface, and the higher spots get a mere vapor of paint over them, which soon fails. At that point, the oxygen now has contact with the steel, and the process starts again. Here is a diagram to illustrate:
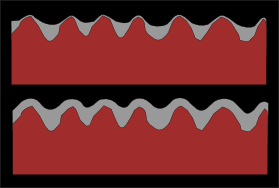 coatings on rust
coatings on rust
The top drawing is a typical finish on rusted metal. The lower one illustrates what happens when a thicker bodied coating is applied. More material stays on the top of the high spots. I don't have microscopic verification of this, however, I was put in a situation that demanded I act on my theory. I was called by Knoxville Locomotive works to paint an older boxcar. Most of the time this boxcar is used for storing parts, but there were times when it might be used as part of an historic charter train, for movie filming and that sort of thing.
I know from extensive experience that G & O pays careful attention to the painting and preservation of equipment that's used regularly, but this storage piece didn't rate that degree of priority. My conversation with the supervisor went like this:
Jim, this is the boxcar that we want put back to its original scheme.
Me: It's solid rust, there might be a total of four square inches that still have paint. It will need to be sandblasted, or hand sanded everywhere, then painted.
Well Jim, it's not in the budget. Can you do anything else?
Me: Hmm, I do have this theory which might solve the problem, let me try it.
I had recently been using a nice product, one which has been very versatile. It's called Devoe (brand) Industrial and tank primer. It's a thick, heavy primer that's in the alkyd (oil based) price range. Yet it can be top coated with urethanes, which have “hotter” (stronger) solvents. Most alkyds can't withstand this treatment. Also, if you let it set for a couple days after applying, it can be sanded in a way similar to a high build sandable primer which auto body technicians use. Their's is more precise, but the price is a lot higher. What I've found is that on many utility projects, like painting welding trailers, This primer can be sanded to a nice enough surface. All in all, I'm really satisfied with this product. It is what I put on the rusted boxcar first. It was rolled on.
A local franchise of a national paint company claimed to be able to make a dark green in oil based paint (budget constraint). They were VERY far off, it turned out to be a light electric lime color. Rather than return it, I told the supervisor That using it as an additional coat of paint would help my theory. So that was the first coat of paint over the primer. The second coat was a color I mixed myself from individual one gallon cans. This was all done five years ago, and upon looking at it a couple months ago, there still was no rust. This boxcar was solid, rough rust. It got no sanding or sandblasting whatsoever. No wire brush, no scraping. You CAN put a lasting finish over very rusted steel, you just need the right products and methods. I highly recommend Devoe's industrial primer.
 Green boxcar
Green boxcar
 link to
link to



